Sarepta Therapeutics Stock Falls as Some Trials Halted After Patient Death

Key Takeaways
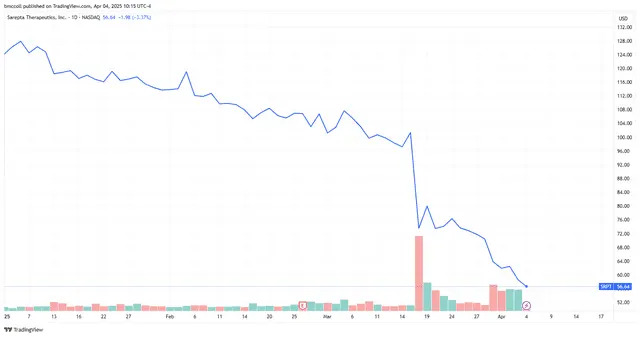
Shares of Sarepta Therapeutics ( SRPT ) slid 6% Friday morning as European regulators called for a pause in some testing of the biopharmaceutical firm's muscular dystrophy drug, Elevidys, following the death of a patient last month.
Sarepta said authorities in European Union (EU) reference member countries "requested that the independent data monitoring committee (DMC) meet to review the adverse event." It added that while that information is finalized, "recruitment and dosing in certain clinical studies of ELEVIDYS are temporarily halted."
The company reported March 18 that a patient with Duchenne muscular dystrophy who was taking Elevidys died of acute liver failure (ALF). Sarepta noted then that "acute liver injury is a known possible side effect of Elevidys" and other similar gene therapies, although this was the first time a death had been reported of anyone taking the drug.
3 Clinical Trials Halted
Three clinical studies are affected by the halt. The company explained that monitoring and data collection for participants already enrolled is continuing, and it does "not anticipate a material impact on the timeline for these studies."
Sarepta Pharmaceuticals shares plunged following the March 18 announcement and now are trading at their lowest level in more than seven years.
Read the original article on Investopedia

